COVID-19
 Recogniti has developed Point-of-Contact Diagnostic of SARS-CoV-2 (COVID-19) capability – a novel diagnostic, surveillance, monitoring and test capability based on existing technology and methods widely used in other domains.
Recogniti has developed Point-of-Contact Diagnostic of SARS-CoV-2 (COVID-19) capability – a novel diagnostic, surveillance, monitoring and test capability based on existing technology and methods widely used in other domains.
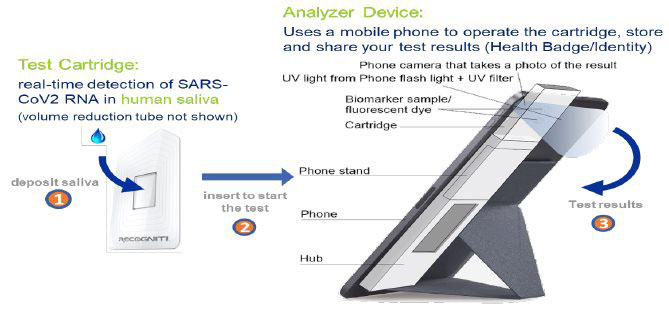
Rapid Autonomous Diagnostics and Awareness based on RNA (RADAR) is an end-to-end consumer-setting solution that helps quarantine and prevent spread of infection through early detection. SARS-CoV-2 detection is based on saliva using loop-mediated isothermal amplification (LAMP) microfluidics assay (Test Cartridge) and an Analyzer Device that operates the Cartridge with a consumer mobile phone.
RADAR does not require any complicated instruments and the test result can be read and displayed on a mobile device. Hence, it can be used at home, airports, metro stations and work spaces, event venues, restaurants or any other Point-of-Entry (PoE) to establish “Health Safe Zones” allowing people to feel protected, as well as employers and businesses to confidently return back to normal. Furthermore, a “Health Badge/Credential” can establish level access for PoE, e.g. office building, store, airplane, taxi, concert event.
Test Cartridge uses three autonomous and integrated stages – 1) Sample collection and preparation; 1-2ml saliva sample is deposited into the sample collection tube, RNA is washed and inserted into the intake channels for stage 2 (macro-to-micro volume reduction). 2) LAMP reaction – working volume in all reaction chambers is 0μL (3x – 5x rescaling may be needed to factor in losses and to lower the manufacturing cost in favor of size); 3) Assessment of Results is then performed by the RADAR analyzer device based on fluorescent light absorbance levels.
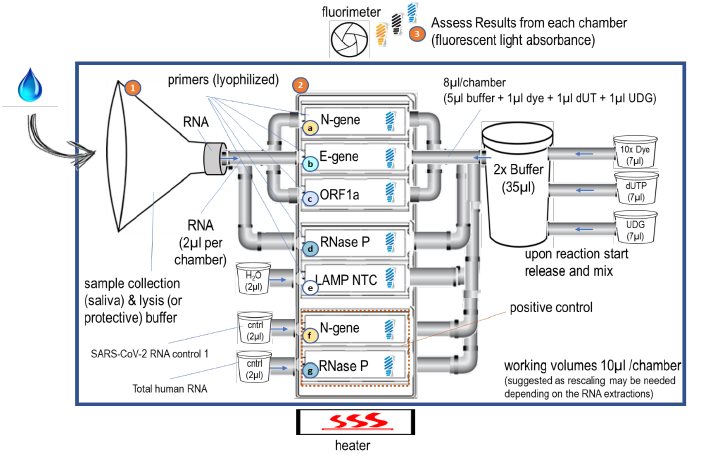
Analyzer Test Device has two primary functions: 1) maintain constant 60-65°C temperature for the operation of the Test Cartridge, and (2) measure once per minute the rate of change of the fluorescent light (430/550 nm) to interpret positive or negative result within each chamber of the Test Cartridge (Figure 1 above) and transmits to the results to the mobile app. Camera flash of a consumer cell phone performs the second function.
RADAR is a “gold standard” RNA test that is already validated.
